by Mark Tiemeyer, MBA, MT(ASCP), Vizient Consulting Director
Walter Vallière, ScD, Vizient Consulting Director
Laboratory testing is an inherent part of the health care system. It’s used across all care settings and specialties for screening, diagnosis and disease management, and has a significant impact on care outcomes and costs. One review of the literature found that laboratory testing is the largest medical activity in terms of volume and may influence more than 70% of medical decision-making.
While hospitals work to improve the health of their patients and communities, they continue to be confronted with financial pressures stemming from the COVID-19 pandemic, such as lost revenue and increased costs associated with labor and treating COVID patients. Hospital laboratory leaders tasked with identifying cost savings may come up short by focusing on traditional, often-used methods such as price reductions, improved contracting and inventory management. Forward thinking laboratory leaders have already accessed these efficiencies.
However, shifting your focus to demand management can be an effective way to secure laboratory cost reductions while supporting physicians, improving outcomes and remaining abreast of emerging science.
Focusing on demand management
Demand management in a hospital laboratory setting is the focus of identifying and curbing inappropriate testing, which can take several forms, including over utilization of tests that are ordered but not indicated. One meta-analysis estimated the rate of inappropriate hospital laboratory testing to be 43.9% at the time of admission and 7.4% for subsequent testing.
Such inappropriate testing utilizes precious labor and supply resources as well increases the potential for false positives and additional interventions which, in turn, can increase costs and reduce patient outcomes.
There are many reasons that inappropriate testing can occur, and a thorough demand management assessment can help identify and address them.
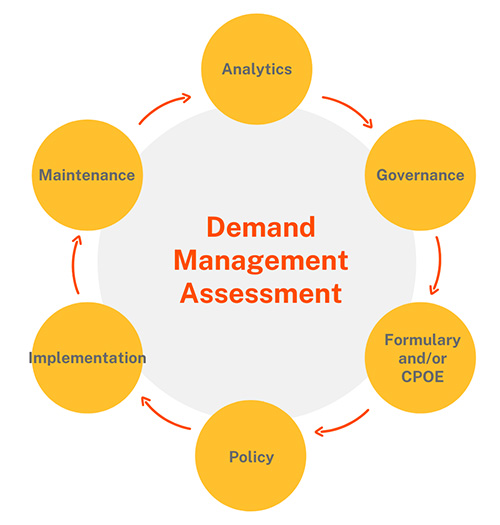
An effective demand management assessment begins with observations and analyses and leads to the development of a formulary or computerized provider order entry (CPOE) of a laboratory’s test offerings with usage rules. The formulary typically eliminates archaic tests, aligns the laboratory’s test offerings with the most recent scientific data and presents medical usage models and process flow charts from scientific data and evidence-based medicine. The goal is to ensure the right test, at the right time, for the right reason.
Hospital laboratory leaders can conduct their own assessment utilizing the steps below or may consider partnering with an outside organization that has the ability to evaluate the latest medical literature and provide an objective assessment.
Analytics—Begin by evaluating which laboratory tests were used during a window of time (one year is a good place to begin). Leverage available sources of such information, such as laboratory information system orders, reference lab usage, HIS reports or Vizient tools.
Evaluate recent scientific methods and clinical practice guidelines to determine tests that may be archaic or underused. Identify trends that you are seeing as well as actions needed to support better operations. For example, do you see that laboratory tests are often ordered the day of a patient discharge despite having a very limited chance of being reviewed?
Governance—Once your analyses have identified areas of focus, a governance structure should be established consisting of physicians and scientists such as chief pathologist/chief science officer, microbiologist, chemist or another scientist. The group discusses the findings and develops recommendations. Common questions the group may explore include: Does the laboratory have the necessary resources and space to run this new test in-house? What will it cost to offer this test? Are there regulatory considerations for this test?
Formulary and/or CPOE—The next step is to develop a list of appropriate laboratory tests and who may order them and include those in the CPOE. You might also consider developing a laboratory formulary, which is analogous to the drug formulary maintained by a hospital pharmacy. It includes what test methodologies will be offered and how they will be delivered, i.e., central testing, near-patient testing, point of care testing or outsourced to an independent reference laboratory. Share the information with your governance group, executive leadership and medical staff, educating them about what was changed and why it was needed.
Policy—In addition to deciding the ‘what’ and ‘how’ of laboratory tests to be offered in the laboratory formulary, policies must be established that define what tests can be ordered and any limitations or restrictions. Given the thousands of laboratory tests available, it is not realistic to expect physicians to stay up to date on the most current recommended ordering practices. Share the policy with the chief financial officer or designee, chief medical officer and other roles that serve as high utilizers of laboratory services.
Implementation—The implementation will vary according to each facility’s governance for introducing changes to test ordering, but communication of the policy will be essential in implementing any proposed changes to the testing policy. Frequently, medical executive committees are engaged to disseminate information, with follow up in specialized areas of focus (cardiology, genetics), and in other physician communications. Support of administration, quality, pharmacy, nursing, laboratory, IT and other staff will be critical.
Maintenance—A regular review of ordering behavior, published literature and professional physician organization recommendations is recommended. Staying abreast of the latest medical laboratory advances optimize the delivery of clinical information to decision makers. The pandemic has revealed the clinical laboratory’s role in the pursuit of faster, more accurate, and better diagnostics in improving patient care.
Understanding and managing demand for hospital laboratory services offers sustainable operational and client service improvements that enhance quality and reduce cost, all while addressing the needs of physicians, technology, and new science. Effectively designed and implemented it represents the combined efforts of physicians and laboratory scientists to offer high-quality laboratory-related tests that are supported by current scientific data and to use these upgraded testing capabilities more effectively.
If you need assistance developing a demand management strategy for your hospital or conducting a demand management assistance, feel free to reach out to us for assistance.
About the authors
Mark Tiemeyer has been a consulting director with Vizient for 12 years. Prior to Vizient, Mark directed contracting activities in laboratory, purchased services and general medical for a large health system for nine years. Mark is a medical technologist by background, having managed and directed hospital and reference labs for over 14 years. Mark brings experience in change management and assembling diverse stakeholders to evaluate, select, negotiate and contract to maximize value for stand-alone hospitals to integrated delivery networks.
Walter Valliere brings more than 49 years of experience leading initiatives to grow market share and reduce operating costs through process improvements, strategic outsourcing, business restructuring, business consolidations, supply chain optimization and new venture development. During 10 years with Vizient, Valliere has also held principal leadership/ownership positions with both a multi-site, multi-state independent laboratory and a specialty consulting firm that served the biotech, biopharma, and health care industries. He also holds certifications in ITIL, PRINCE2 project management, and Six Sigma.
