In the dynamic world of spine and neuromodulation devices, it’s more important than ever for non-clinical hospital staff to stay informed on market trends and new devices entering the market. In spine, the number of “me too” devices and lack of clinical data make decision-making complex.
Understanding the what, how and when of the products being developed allows health care organizations to more effectively plan purchases and guide clinical practice. Vizient’s semiannual Technology Watch is a resource for service line and supply chain professionals to get the information they need to talk with physicians and clinicians about current and new products.
The spine market has always been a dynamic environment with open access for new suppliers with new ideas. The U.S. FDA 510k clearance process, which requires little clinical evidence of unique product benefit, has led to numerous suppliers entering the market with like products yet at greatly varying prices.
“Because of the market dynamics, health care organizations struggle to gain control of their spine spend while also delivering reproducible patient care and quality outcomes,” said Craig Lukowski, director, physician preference contract services. “In the emerging pay-for-value system, reduction of suppliers will be one of the factors required to improve quality outcomes and lower cost.”
The full report includes pricing trends, supplier updates and insights into the most recent findings from Vizient orthopedic experts and can be downloaded here. Following are the key takeaways:
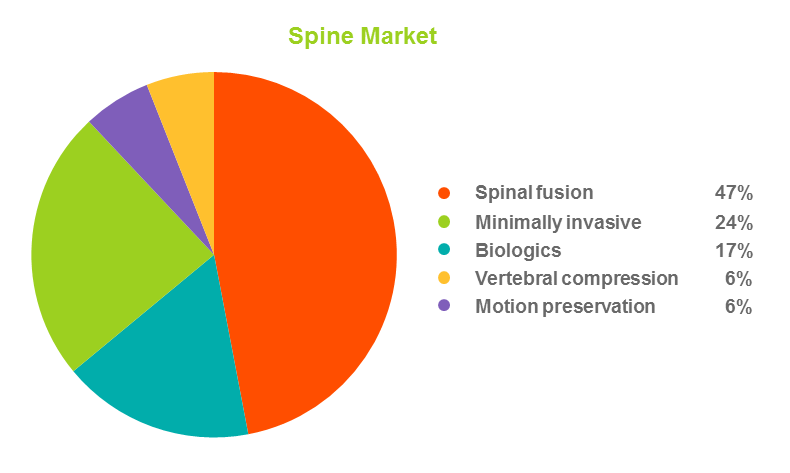
1. Spine market shows steady growth. The U.S. spine market is now estimated to equal the size of the U.S. total joints market. According to the Becker Spine Report, the estimated value of the spine surgery market is more than $7 billion and is expected to reach $11 billion by the end of 2020.
New motion-preserving devices are being developed, which may help improve patient care, but also may begin to moderate pricing. Several manufacturers in the spine marketplace are now using 3D printing, which offers enhanced design capability, enabling the development of implants that enhance bone ingrowth and the potential to create implants customized to the patient.
2. Neuromodulation rapidly expanding in procedures and uses. This complex therapy, which is characterized by an implantable device that applies low-level energy to the nervous system to block nerve signals, is projected to grow at double digit rates through 2020.
During the past year, several new neuromodulation devices have been approved for both traditional and new indications. The accelerated cost and emerging applications for these devices is creating cost concerns for hospitals as the devices continue to erode thin reimbursement returns. Strong physician preference and supplier relationships make pricing control very challenging.
“The neuromodulation market, while providing therapies that improve patient care options, creates a scenario that could lead to negative supplier practices, like increasing prices and exceptionally strong physician supplier relationships.” said Lukowski. “It’s a mixed blessing.”
As health care continues its rapid change, it is crucial for hospitals to be well informed about new devices that will affect both the care they provide and their bottom line. Excitement and caution are the key words for these markets.